We talk a lot about manuscript structure and writing, but figures (and tables) are just as important for communicating your research. Figures can include graphs, photographs, drawings, medical scans, schematics, and charts. (In publishing, all illustrations are usually called “figures.”)
Figures help you present observations or a large amount of data quickly and efficiently, and they catch the reader’s attention. Busy readers who want to quickly assess an article often check the title, abstract, and figures before anything else. That’s because the figures often show the most important results.
Your figures therefore need to be well-prepared, easy to read, and informative. You also need to be sure they follow the guidelines (journal or other) and they’re presented with suitable permissions. We’ll go over all that here.
What you’ll learn in this post
• What to put in the figures in your scientific research.
• Choosing and presenting the data effectively in your figures.
• A checklist of key items to watch for when you create and publish your figures.
Choosing the right data for your figures
Journals often have a limit on how many figures you can include in the main manuscript, so choose your figures wisely. Focus on the user/reader experience.
Think about what readers or an editor might want to see as a figure when they’re reading. Think about:
- What are your most important results?
- Do you have images that show your findings unambiguously and that will help readers understand them?
- What patterns are in the data that could be shown graphically to add to your argument?
- What data would be easier to explain in a figure then in writing?
Elements of a good figure
Once you’ve selected the data you’ll use for a figure, you need to consider the best way to show it. For a rundown of choosing software for designing your figures, read this Learning Lab article.
Making a clear message
Several aspects are key in whether you communicate the information effectively in a figure
- Clear labeling
- Simple presentation
- Highlighted points of interest
These will all improve the reader’s experience and let them understand your data more easily than if they were reading about it in words.
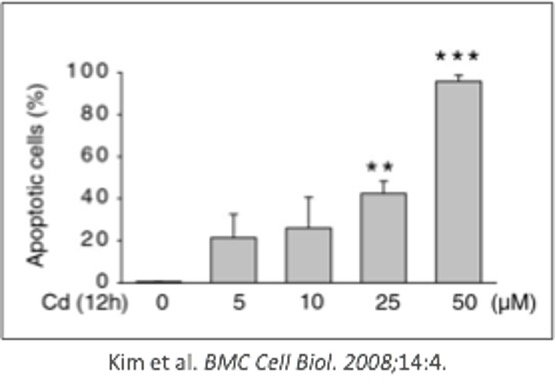
For example, in the chart above, the authors show a simple pattern of increasing percentage of apoptotic cells with increasing amounts of cadmium.
The axes are clearly labeled, including the units, and the data points are simple and easy to interpret.
The authors also indicate changes that are statistically significant with asterisks (*), which would be defined in the figure legend.
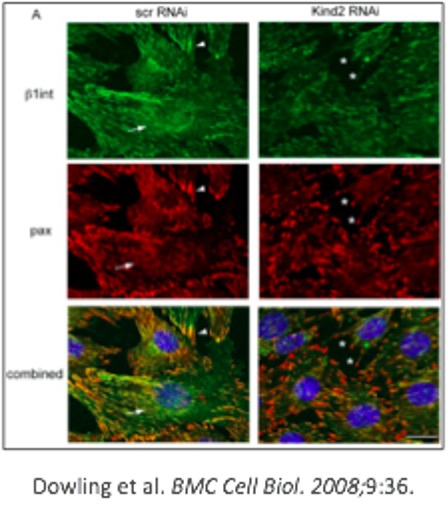
Kindlin-2 knockdown and focal adhesion localization. A. Confocal immunofluorescent microscopy with anti-β1 integrin (green) and anti-paxillin (red) on C2C12 cells transfected with RNAi and then changed to differentiation media for 2 days. Control cells (scr RNAi) show linear staining consistent with localization to costameres (arrows), as well as punctate focal contact staining (arrowheads). Conversely, focal contact proteins in the kindlin-2 RNAi cells fail to form linear structures and instead are concentrated in unusual appearing puncta (*). (Scale bar = 20 μM).
Similarly, the image above is easy to interpret thanks to several key elements.
First, the figure has a title that clearly states what this image is showing. The authors then briefly describe the essential methods relevant to the figure; in this case, what stains were used and which colors represent them.
The main results are then stated, and any key points of note in the image are labeled with indicators, which are also defined in the legend.
Get your scaling right
When presenting images, it’s also important to indicate the scale. You can do this either in the legend or in the figure itself. This way readers now have all the essential information to properly assess and interpret the findings.
Checklist for display items
- Stand-alone legends
- Comply with the journal’s specified number of display items
- Avoid redundancy among display items or between display items and text
- Divide data showing different effects or parameters among different panels within the same display item
- Use scale bars, trend lines and clear labels, and show the results of statistical tests
- Avoid or define all symbols and abbreviations
- With large samples, show % changes/differences as well as absolute values
- Submit figures in a separate file or at the end of the manuscript file rather than embedded in the main text
- Check the instructions for authors for any specific requirements regarding format, size, color, number of items and any other parameters
Wrapping it up
Once you’ve made your images, take a step back and put yourself in the reader’s shoes. If you were unfamiliar with this study, would there be any details missing for you to understand it? Good figures motivate readers to read your full manuscript, and maybe even lead to increased citations.
So don’t miss your chance to catch someone’s eye.